New Research, More Doubts about Tubing Safety for Infants
December 18, 2019
Certain hospital tubing devices could actually endanger infants, more new research confirms.
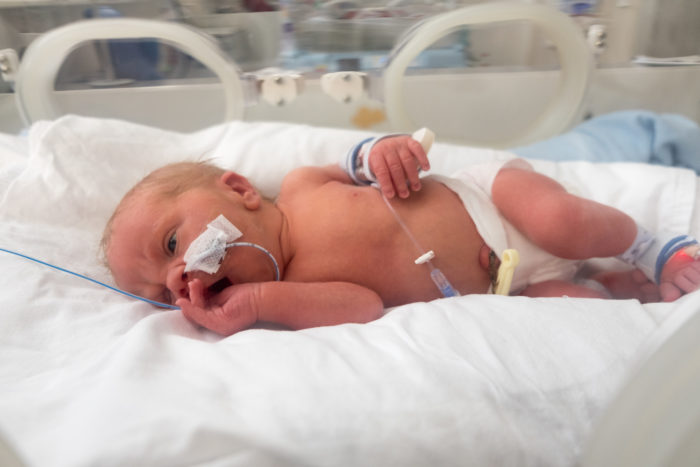
A recent University of Florida study found that the risk of inaccurate dosing with new tubing technology known as ENFit is significantly higher than accepted standards. ENFit was designed to reduce misconnection errors through a system in which tubes have unique shapes and functions – making them easier to distinguish from one another. But ENFit’s design introduces a new risk. The connector features a moat where excess medication can linger. That means syringes can deliver the wrong amount of medication.
For infants, this can be deadly. Precision is critical for small doses – and small patients. Perhaps that’s why ENFit has sparked concerns before. Just earlier this year, a separate study demonstrated that the ENFit tubing connector “significantly increases the opportunity for inaccurate dosing.”
As an example of potential dosing dangers, University of Florida researcher and pharmacist Keliana O’Mara alludes to infants facing neonatal abstinence syndrome. An infant being weaned off of opioids might receive morphine every three hours for 30 days. “That’s 240 chances to have a problem with dosing accuracy,” O’Mara emphasized.
Safety issues also raise difficult questions for hospitals. While some have voluntarily transitioned to using ENFit, other hospitals are resistant to the transition, citing patient safety risks and lack of evidence. Other safe, FDA-approved options continue to be available.
Finding the right balance between innovation and safety can be challenging. As hospitals consider new tubing technology, they should move cautiously and with the benefit of all available information. The needs of the tiniest patients depend upon it.
Categorized in: Blog

